
DCMS online
. org
Northeast Florida Medicine
Vol. 66, No. 4 2015
41
Inflammatory Bowel Disease
in certain tissues. Autoimmune attack can happen many
years after the removal of the colon.
3
In the case of primary sclerosing cholangitis, it is sus-
pected that memory lymphocytes that have been primed in
the bowel can recirculate for many years after the removal
of the colon. This can cause damage until the occurrence
of a stimulus in the liver that activates inflammation and
overexpression of the adhesion molecules and persistent
lymphocyte recruitment. Interference with adhesionmole-
cules may be very useful in the treatment of extraintestinal
manifestations of IBD.
The high prevalence of p-ANCA in patients with UC,
commonly associated with PSC, Erythema nodosum (EN)
and uveitis, supports the role of autoimmune mechanisms
in the development of EIM. (Table 1)
There appears to be great genetic susceptibility with
EIMs in inflammatory bowel disease. These EIMs have a
familial predisposition seen in 83 percent of concordance
among siblings, and there appears to be a strong genetic
influence leading to the identification of many suspected
predisposal type genes.
3
The Human Leukocyte Antigen
(HLA) system is considered one of the major genetic mark-
ers associated with EIMs in IBD. It has been very clearly
reported that UC patients that have HLA-B8 or the DR3
phenotype have a tenfold higher risk of primary sclerosing
cholangitis.
4
We see a variety of other HLA phenotypes that
predispose people to ocular and articular manifestations,
especially patients with the HLA-B27 and B58 phenotypes
who also have a higher risk of uveitis. HLA-B27 is strongly
associated (up to 80 percent) with ankylosing spondylitis.
2
Polymorphisms of alpha-TNF have been associated with
erythema nodosum in IBD patients. Polymorphisms of
NOD2 (Card15) are associated with familial Crohn’s, ileal
disease, fibrostenotic Crohn’s, and sacroiliitis.
2
Table 1. Pathogenesis of EIM in IBD
- Genetic susceptibility
- Antigenic display of autoantigen
- Aberrant self-recognition
- Immunopathogenetic autoantibodies (anti-tropomysin)
- Immune complex formation
- Cytokine imbalances
- Bacterial antigens or toxins
In the past decade, we have seen the concept of dysbiosis
of the gut microbiome emerge as a potential pathogenetic
focus in IBD. The judicious use of antibiotics, probiotics,
prebiotics, enteral nutrition, and fecal transplantation
are flourishing.
Arthropathies
The reported incidence of arthropathies associated with
IBD range from 4 to 23 percent.
5
Two types have been
defined and the distinction is supported by differences in
genetic susceptibility. Type 1 is a large joint pauciarticular
arthropathy that occurs at times of IBD activity. Type 2
is polyarticular small joint (frequently 5 or more joints)
arthropathy where activity is largely independent of IBD
activity. Axial arthritis includes sacroiliitis and ankylosing
spondylitis which have characteristic radiologic changes.
(Figure 1) HLA B-27 is overrepresented in axial arthritis
related to IBD.
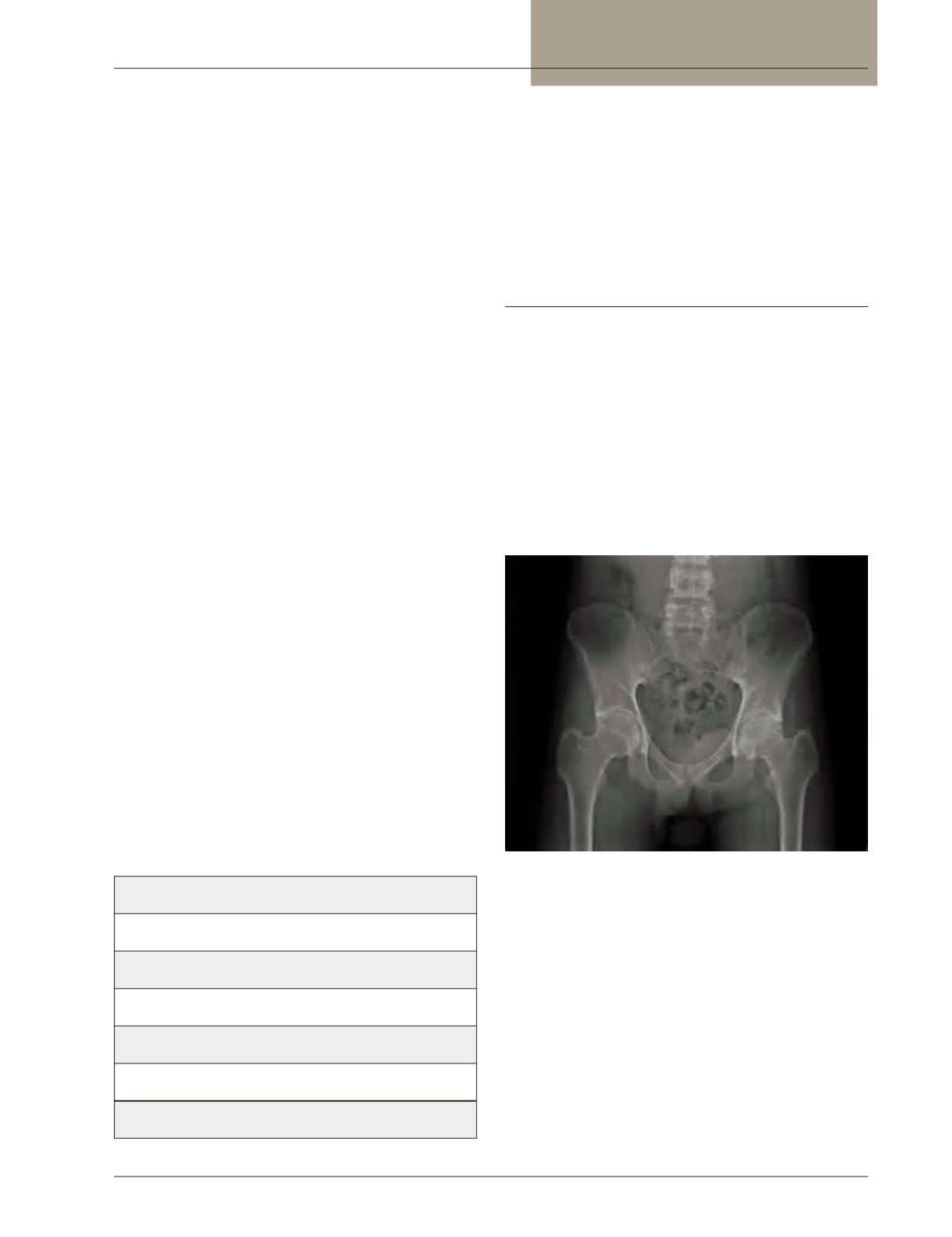
Figure 1:
Sacroiliitis in Crohn’s disease
Type 1 arthropathy affects the weight-bearing joints
including the ankles, knees, hips, wrists, elbows, and
shoulders. Pauciarticular refers to fewer than 5 joints being
affected. The arthritis is usually acute and self-limiting,
and usually resolves within a few weeks as the disease it-
self decreases. It typically does not leave permanent joint
damage. These joints are painful, tender, and swollen. The
differential diagnosis could include osteoarthritis, septic ar-
thritis, pseudogout, and, occasionally, rheumatoid arthritis.
The polyarticular peripheral arthropathy, or type 2,
affects the small joints of the hands as a symmetrical
















